ڤيتامين د
| ڤيتامين د | |
|---|---|
| صف عقاقير | |
 الكولـِكالسيفـِرول (د3) | |
| مميزات الصف | |
| الاستخدام | الرخد، هشاشة العظام، نقص ڤيتامين د |
| كود ATC | A11CC |
| الهدف الحيوي | مستقبل ڤيتامين د |
| البيانات الإكلينيكية | |
| Drugs.com | MedFacts Natural Products |
| الوصلات الخارجية | |
| MeSH | D014807 |
| In Wikidata | |
ڤيتامين د (إنگليزية: Vitamin D)،،هو منظّم الجسم الأساسي لتوازن الكالسيوم. يساعد على تزويد العظم بالمعادن وتطوير الهيكل العظمي. يعتبر مؤشر هورموني، وليس له ذاته أي نشاط هورموني، مع أنه من المكن أن يتم تحويله إلى جزيئة ذات نشاط هورموني.
تتضمن كلمة ڤيتامين د عددا من 1 حتى 5 وهي مواد ثابتة حرارياً ومن أهم هذه الڤيتامينات ڤيتامين د2 المسمى إرگوكالسيفـِرول وينتج بتأثير الاشعة فوق البنفسجية على الستيرول النباتي والآخر ڤيتامين د3 والمسمى كولـِكالسيفـِرول: وهو المركب الطبيعي المضاد للكساح وينتج بتاثير الاشعاع على ڤيتامين أولي أو پروڤيتامين ديهدروكولسترول-7 الموجود في الجلد.
المصدر الطبيعي الرئيسي للڤيتامين هو تخليق الكولـِكالسيفـِرول في الطبقات السفلية من البشرة من خلال تفاعل كيميائي يعتمد على التعرض لضوء الشمس (خاصة الأشعة فوق البنفسجية).[1][2] يمكن الحصول على الكولـِكالسيفـِرول والإرگوكالسيفـِرول من النظام الخذائي ومن المكملات.[3][4][5] لا يوجد سوى عدد قليل من الأطعمة، مثل لحم الأسماك الدهنية، تحتوي بشكل طبيعي على كميات كبيرة من ڤيتامين د.[6][7] في الولايات المتحدة ودول أخرى، يتم تعزيز حليب البقر وبدائل الحليب المشتق من النباتات بڤيتامين د، وكذلك العديد من أنواع حبوب الإفطار. تعرض الفطر للأشعة فوق البنفسجية يزودها بكميات مفيدة من ڤيتامين د.[6] عادةً ما تفترض التوصيات الغذائية أن ڤيتامين (د) يتناوله الأفراد عن طريق الفم، نظرًا لأن التعرض لأشعة الشمس بين السكان متغير والتوصيات بشأن مقدار التعرض الآمن للشمس غير مؤكدة نظرًا لخطر الإصابة بسرطان الجلد.[6]
ڤيتامين د من النظام الغذائي، أو من تخليق الجلد، غير نشط بيولوجياً. يجب على إنزيم پروتين الهيدروكسيلات تحويله إلى الشكل النشط. يحدث ذلك في الكبد والكلى. نظرًا لأنه يمكن تخليق ڤيتامين د بكميات كافية من قبل معظم الثدييات في حالة تعرضها لأشعة الشمس الكافية، فإنه ليس ضروريًا، لذلك من الناحية الفنية ليس ڤيتامين.[5] بدلاً من ذلك، يمكن اعتباره هرموناً، مع تنشيط هرمون ڤيتامين د الذي ينتج عنه الشكل النشط، الكالسيترول، والذي ينتج بعد ذلك تأثيرات عبر المستقبلات النووية في مواقع متعددة.[5] يتحول الكولـِكالسيفـِرول في الكبد إلى كالسيفديول (25-هيدروكسي كولـِكالسيفـِرول)؛ يتم تحويل إرگوكالسيفـِرول إلى 25 هيدروكسي إرگوكالسيفـِرول. يُقاس مستقلب ڤيتامين د (يسمى 25-هيدروكسي ڤيتامين د أو 25(OH)D) في مصل الدم لتحديد حالة ڤيتامين د في الجسم.[8][9] يتم هيدروكسلة الكالسيفديول عن طريق الكلى لتكوين الكالسيتريول (المعروف أيضًا باسم 1،25-ديهيدروكسي كولـِكالسيفـِرول)، وهو الشكل النشط بيولوجياً من ڤيتامين د.[10] يدور في الدم الكالسيتريول كهرمون، وله دور رئيسي في تنظيم تركيز الكالسيوم والفوسفات، وتعزيز النمو الصحي وإعادة تشكيل العظام. يحتوي الكالسيتريول أيضًا على تأثيرات أخرى، بما في ذلك بعض التأثيرات على نمو الخلايا والوظائف العصبية العضلية والجهاز المناعي وتقليل الالتهاب.[6]
لڤيتامين د دوراً هاماً في تخليق الكالسيوم وأيضه. كان اكتشافه نتيجة جهد للعثور على المادة الغذائية التي يفتقر إليها الأطفال المصابون بالكساح (شكل من لين العظام في مرحلة الطفولة).[11] يتم إعطاء مكملات ڤيتامين د للعلاج أو للوقاية من لين العظام والكساح. الأدلة على الآثار الصحية الأخرى لمكملات ڤيتامين د في عموم السكان غير متسقة.[12][13] إن تأثير مكملات ڤيتامين د على الوفيات غير واضح، حيث وجد تحليل تلوي انخفاضًا طفيفًا في معدل الوفيات لدى كبار السن،[14] وخلص آخر إلى عدم وجود مبرر واضح للتوصية بمكملات للوقاية من العديد من الأمراض، وأنه ليس هناك حاجة لمزيد من البحث لتصميم مماثل في هذه المجالات.[15]
الأنواع
| الاسم | التركيب الكيميائي | البنية |
|---|---|---|
| ڤيتامين د1 | خليط من المركبات الجزئيية الإرگوكالسيفـِرول واللوميسترول، 1:1 | |
| ڤيتامين د2 | الإرگوكالسيفـِرول (محضر من الإرگوسترول) | 
|
| ڤيتامين د3 | الكولـِكالسيفـِرول (محضر من 7-ديهيدروكولسترول في الجلد). | 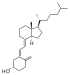
|
| ڤيتامين د4 | 22-ديهيدروكسيإرگوكالسيفـِرول | 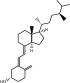
|
| ڤيتامين د5 | السيتوكالسيفرول (محضر من 7-ديهيدروسيتوسرول) | 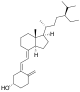
|
هناك عدد من أشكال (أشباه الڤيتامينات) ڤيتامين دول. الشكلان الرئيسيان هما ڤيتامين D2 أو الإرگوكالسيفـِرول، وڤيتامين د3 أو الكولـِكالسيفـِرول؛ ڤيتامين د بدون الإشارة إلى ڤيتامين د2 or د3 أو كلاهما. تُعرف هذه الأنواع مجتمع باسم الكالسيفرول.[16] تم التعرف على ڤيتامين د2 كيميائياً عام 1931. وفي عام 1935، عُرف التركيب الكيميائي لڤيتامين د3 وأُثبت أنه يُخلق نتيجة تعرض 7-ديهيدروكولسترول للأشعة الفوق بفنسجية.[17]
كيميائياً، هناك أشخاص مختلفة من ڤيتامين دا، وهي السيوسترويدات، مثل السترويدات حيث يتم كسر إحدى الروابط الموجودة في حلقات السترويد.[17] الاختلاف في التركيب بين ڤيتامين د2 وڤيتامين د3 هو السلسلة الجانبية في د2 والتي تحتوي على رابطة مزدوجة بين كربون 22 و23، ومجموعة ميثيل على كربون 24.
إمتصاصه
يمتص ڤيتامين د في النصف الأعلى من الامعاء الدقيقة في وجود املاح الصفراء وكذلك يرتبط بالالفا -جلوبيولين في البلازما. من أهم مناطق تخزين الڤيتامين في الجسم: العضلات والنسيج الدهني.
استعمالاته
يساعد ڤيتامين د في: تشكيل خلايا الدم، المناعة، تمايز الخلايا الأمرالذي قد يقلل من أخطار السرطان. أظهر ڤيتامين د قدرته على توفير الحماية من أمراض المناعة كالتهاب المفاصل المناعي، تصلّب الأنسجة المتعدّد، وسكّري الأطفال.
يساعد ڤيتامين د الجسم على الحفاظ على مستويات الأنسولين الضرورية في الدم. توجد مستقبلات ڤيتامين د في البنكرياس، حيث يتم إنتاج الأنسيولين. كبسولات البدائل قد تزيد من إفراز الإنسولين في الأشخاص المصابين بالسكّري النوع 2.
ڤيتامين د يستعمل في الحالات التالية:
- - مرض كرون
- - التليف الكيسي
- - ترقق العظام
- - داء الكساح .
إن المدى الذي يساهم به ڤيتامين د للمساعدة على تخفيض الكسور وخسارة العظم لدى المسنين ما زال غير مؤكّدا، على أية حال بعض الدراسات تشير إلى أنه مفيد في هذه الحالات.
أبحاث متقدمة تشير إلى إمكانية استعمال ڤيتامين د أيضا للمساعدة في الكآبة الموسميّة، الطرش القوقعي الثنائي، مقاومة زيادة النوبات، وتساعد بتصلّب الأنسجة المتعدّد ومتلازمة الجيب المريضة.
الكيمياء الحيوية
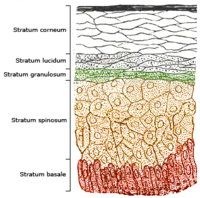
انتاجه في الجلد
عند التعرض للشمس يتحول ديهيدروكولسترول بوجود الأشعة فوق البنفسجية إلى ڤيتامين د.
ڤيتامين د كڤيتامين
حيث أن فيتامين د يُنتج طبيعياً في الجسم البشري لكنه يصبح فيتاميناً فقط بسبب التغيرات الثقافية، المرتبطة بالحضارة والتي قللت الإنتاج الطبيعي بسبب قلة تعرض جلد الإنسان للشمس، وبسبب استخدام المزيد من الملابس والخروج في الهواء الطلق لوقت أقل.[18]
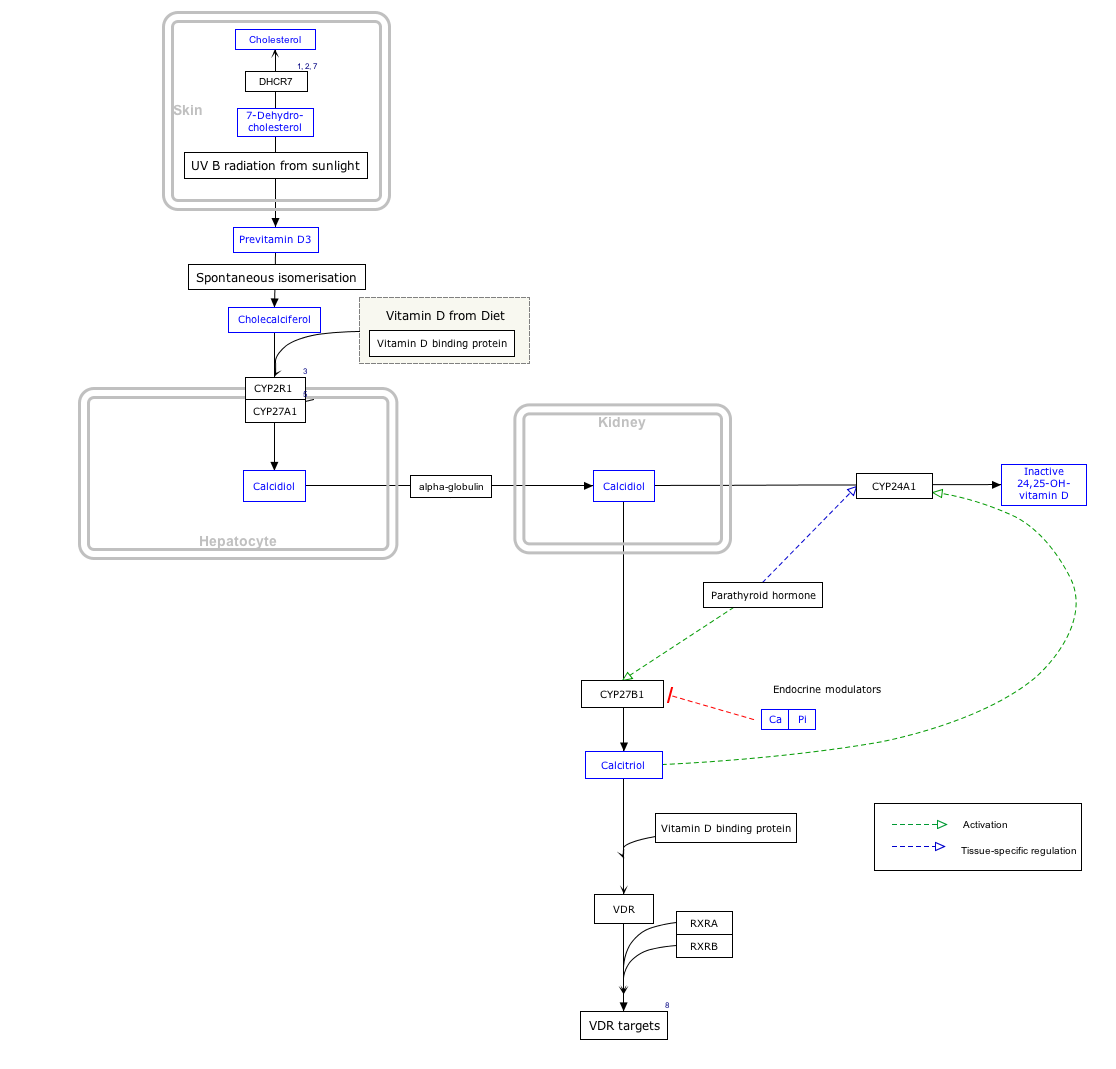
آلية التخليق (الشكل 3)
| 1. ڤيتامين د3 is synthesized from 7-dehydrocholesterol, a derivative of cholesterol, which is then photolyzed by ultraviolet light in 6-electron conrotatory electrocyclic reaction. The product is pre-vitamin D3. |
 |
| 2. Pre-vitamin D3 then spontaneously isomerizes to Vitamin D3 in a antarafacial hydride [1,7]Sigmatropic shift. |
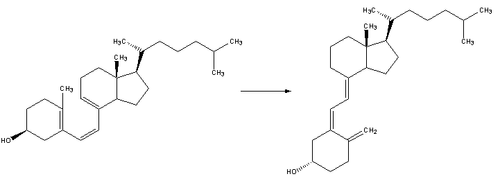 |
| 3. Whether it is made in the skin or ingested, vitamin D3 (cholecalciferol) is then hydroxylated in the liver to 25-hydroxycholecalciferol (25(OH)D3 or calcidiol) by the enzyme 25-hydroxylase produced by hepatocytes, and stored until it is needed.
25-hydroxycholecalciferol is further hydroxylated in the kidneys by the enzyme 1α-hydroxylase, into two dihydroxylated metabolites, the main biologically active hormone 1,25-dihydroxycholecalciferol (1,25(OH)2D3 or calcitriol) and 24R,25(OH)2D3. This conversion occurs in a tightly regulated fashion. Calcitriol is represented below right (hydroxylated Carbon 1 is on the lower ring at right, hydroxylated Carbon 25 is at the upper right end). |
 |
آلية العمل
نقص ڤيتامين د

نقص ڤيتامين د في الأطفال قد يسبّب،الكساح (Rickets) والتي تتميز باضطراب في تكوين العظام نابعا من نقص الكالسيوم والفسفور في النهايات الغضروفية للعظام.كذلك يسبب نقص هذا الڤيتامين في البالغين لين وترقق عظام (osteomalacia) هذه الحالة تعالج عادة بمعوِّضات الكالسيوم وڤيتامين د. إن ترقق العظام يجب أن يشخّص ويعالج من قبل طبيب مختص. قد تحدث تشكيلات عظمية شاذّة في أولئك المصابين بنقص ڤيتامين د. يكون النقص أكثر شيوعا في الشتاء حيث الحصول على نور الشمس صعب. حالات نقص ڤيتامين د شائعة أيضا في المناطق الملوّثة جدا، حيث يمكن أن تُحجب الأشعة فوق البنفسجية.
النباتيون الصارمون، مدمنو الخمور، أولئك المصابون بأمراض الكبد أو الكلية، والأشخاص ذو البشرة السمراء، معرضون أكثر لنقص ڤيتامين د. المصابون بأمراض الكبد أو الكلية ينتجون ڤيتامين د لكن لا ينشّطونه.
الأفرادالذين يعانون من سوء الامتصاص المعوي عموما لديهم نقص ڤيتامين د. أولئك الذين تكون وظيفة البنكرياس عندهم غير كافية، مثل حالات التليف الكيسي أو التهاب البنكرياس، أو مصابون بفرط الدرقية، في أغلب الأحيان لديهم نقص ڤيتامين د.
دوره في العلاج والوقاية من السرطان
يقول علماء ان استخدام جرعات كبيرة من ڤيتامين د يمكن أن تخفض الى النصف مخاطر التعرض للاصابة بالسرطان. وقام باحثون بمراجعة 63 دراسة قديمة وخلصوا الى أن ڤيتامين د يمكن ان يخفض مخاطر تطور سرطان الثدي والمبايض والقولون وانواع اخرى من السرطان.
وقد أجريت هذه الدراسات ونشرت ما بين أعوام 1966 و2004 ، وهي تشمل 30 دراسة بحثية حول سرطان القولون و13 حول سرطان الثدي، و26 حول سرطان البروستاتا و7 حول سرطان المبايض.
وقال الباحثون على الاثر أن التحليلات تشير الى انه في انواع معينة من السرطان فانه لا يمكن تجاهل دور ڤيتامين د.
ترحيب حذر
رحبت الجمعيات الخيرية بحذر بالدراسة التي أجرتها جامعة كاليفورينا، الا انها نبهت إلى أن الإفراط في استخدام ڤيتامين د يمكن أن يلحق أضرارا بالكلى والكبد.
وينتج الشكل الطبيعي من "ڤيتامين د" من الجلد عقب التعرض لاشعة الشمس ويطلق عليه "دي -3"، ويمكن الحصول على الڤيتامين ايضا من بعض الاطعمة مثل الاسماك الغنية بالزيت واللحوم. ورصدت الدراسة التي اجريت في جامعة كاليفورينا بسان دييغو العلاقة بين تناول"ڤيتامين د" وانخفاض معدلات الاصابة بالسرطان.
وفي هذا السياق اشارت الدراسة الى تزايد معدلات الاصابة بالسرطان بين الافارقة والكاريبيين مقابل انخفاضها لدى البيض ، ربما بسبب ان البشرة السوداء لا تنتج كمية كافية من"ڤيتامين د".
ويقول العلماء ان تناول 25 ميلجرام من "ڤيتامين د" يوميا يمكن ان يقلل خطر الاصابة بسرطان القولون بنسبة 50 بالمئة وخطرة الاصابة بسرطان الثدى والمبايض بنسبة 30 بالمئة. إلا ان العلماء اقروا بأن تناول كميات اكبر من "ڤيتامين د" هو امر يجب التعامل معه بحذر.
يقول العلماء ان تناول خمسين ميلجرام من "ڤيتامين د" قد يؤدي إلى أن الجسم قد يمتص كمية كبيرة من الكالسيون وهو امر قد يتسبب في الاضرار بالكبد والكلى.
ويرى البروفيسور سيدريك جارلاند الذي ترأس فريق البحث أنه تبين أن هناك حاجة لايلاء "ڤيتامين د" عناية أكبر.
ويعتبر جارلاند أنه في حال عدم وجود أشعة شمس كافية، فانه يمكن الحصول على الڤيتامين من مصادر أخرى، غير أنه اشار أيضا إلى أن التعرض للشمس له محاذير اخرى.
ويضيف على ذوي البشرة الداكنة التعرض بشكل أكبر لاشعة الشمس من اجل الحصول على كمية كافية من ڤيتامين د.
لا أدلة
بيد ان البروفيسور كولين كووبر من معهد ابحاث السرطان فيقول ان هناك حاجة لاجراء المزيد من الابحاث للحصول على ادلة مؤكدة بشأن فوائد "ڤيتامين د". ويقول كووبر ان الدراسة اخفقت في تقديم أي آلية بشان الكمية المطلوبة من ڤيتامين د التي تقي من السرطان. [19]
سرطان البروستاتا
في محاولة جديدة وجادة لعلاج أكثر أنواع السرطانات الذكورية شيوعا في العالم ، نجح باحثون أمريكيون في تطوير أقراص بڤيتامين " د " لمعالجة المصابين بسرطان البروستاتا في حالة متقدمة. وقد تمكنت شركة صناعة الأدوية بالولايات المتحدة "نوفاسيا" من تصنيع حبوب دواء تحتوي على هذا الڤيتامين بصورة مركزة، دون مخاطر الأعراض الجانبية المترتبة عن جرعة مُفرطة .
ومن المتوقع أن يكون العقار "Asentar DN-101" متوفرا قبل عام 2009، إذا ما نجحت الاختبارات الطبية ، حيث سيُعطى هذا الدواء للمصابين الذين بلغوا مرحلة متقدمة من المرض، بالإضافة إلى العلاج الكيميائي.
أوضح أحد الباحثين بجامعة بيرمنجهام، أن العقار أدى إلى نتائج مثيرة في المراحل الأولى من اختبارين طبيين ، مؤكدا أن الذين تناولوا هذا الدواء ارتفع معدل أمل الحياة لديهم بحوالي تسعة أشهر، مقارنة مع مرضى آخرين لم يتناولوا سوى عقار العلاج الكيماوي تاكسوتير.
وينتظر أن يُنصح المصابون بتناول قرص واحد كل أسبوع، إلى جانب تتبع نظام التاكسوتير الأسبوعي الذي يمتد على ثلاثة أسابيع من أصل أربعة.
جدير بالذكر أن سرطان البروستاتا ينتشر في المناطق البعيدة عن خط الاستواء، بسبب قلة ضوء الشمس. [20]
وفيات السرطان
أظهرت نتائج دراسة طبية، أُجريت على عينة من المرضى في ألمانياً، نُشرت في 4 فبراير 2021، انخفاضًا كبيرًا في معدل وفيات السرطان بسبب تناول مكملات ڤيتامين د، لمن هم فوق سن الخمسين، بنسبة 13%، أي بحوالي 30.000 حالة وفاة سنوياً. كما ينعكس هذا على التكاليف التي تُخصص لرعاية مرضى السرطان في مراحله الأخيرة (900 مليون يورو سنوياً).[21]
الجرعة الموصّى بها
ڤيتامين د الذي يكون على شكل مكملات (كبسولات) يمكن الحصول عليه كڤيتامين د 2 -إرجوكالسيفيرول أو كڤيتامين د 3 -كوليكالسيفيرول. تحتوي الكبسولات متعددة الڤيتامين في العادة على جرعة ڤيتامين د الموصّى بها عموما 200-400 آي يو (وحدة دولية)، أو 5-10 ميكروغرام، كلّ يوم. 400 آي يو توجد في حبوب ڤيتامينات ما قبل وبعد الولادة.
المسنون والمصابون بمتلازمات سوء الامتصاص، والفشل الكبدي، ومتلازمة الالتهاب الكلوي، يأخذون مكملات تحوي على 50,000 آي يو، أو 1,250 ميكروجرام، إسبوعيا لفترة ثمانية أسبابيع. هذه العلاجات يجب أن ترتّب تحت إشراف الطبيب.
الحصة الغذائية
المستويات الموصى بها
| الولايات المتحدة | ||
| المجموعة العمرية | RDA (و.د./يومياً) | (μg/يومياً)[22] |
|---|---|---|
| الرضع 0–6 شهر | 400* | 10 |
| الرضع 6–12 شهر | 400* | 10 |
| 1–70 سنة | 600 | 15 |
| 71+ سنة | 800 | 20 |
| الحامل/المرضعة | 600 | 15 |
| المجموعة العمرية | الحصة القصوى المحتملة (و.د./يومياً) | (µg/يومياً) |
| الرضع 0–6 شهر | 1.000 | 25 |
| الرضع 6–12 شهر | 1.500 | 37.5 |
| 1–3 سنة | 2.500 | 62.5 |
| 4–8 سنة | 3.000 | 75 |
| 9+ سنة | 4.000 | 100 |
| الحامل/المرضعة | 4.000 | 100 [22] |
| كندا | ||
| المجموعة العمرية | RDA (و.د.) | الحصة القصوى المحتملة (و.د.)[23] |
| الرضع 0–6 شهر | 400* | 1.000 |
| الرضع 7–12 شهر | 400* | 1.500 |
| الأطفال 1–3 سنة | 600 | 2.500 |
| الأطفال 4–8 سنة | 600 | 3.000 |
| الأطفال والبالغون 9–70 سنة | 600 | 4.000 |
| البالغون> 70 سنة | 800 | 4.000 |
| الحمل والرضاة | 600 | 4.000 |
| أستراليا ونيوزيلندا | ||
| المجموعة العمرية | الحصة الكافية (μg) | الحصة القصوى (μg)[24] |
| الرضع 0–12 شهر | 5* | 25 |
| الأطفال 1–18 سنة | 5* | 80 |
| البالغون 19–50 سنة | 5* | 80 |
| البالغون 51–70 سنة | 10* | 80 |
| البالغون > 70 سنة | 15* | 80 |
| الهيئة الأوروپية لسلامة الغذاء | ||
| المجموعة العمرية | الحصة الكافية (μg)[25] | الحصة القصوى المحتملة (μg)[26] |
| الرضع 0–12 شهر | 10 | 25 |
| الأطفال 1–10 سنة | 15 | 50 |
| الأطفال 11–17 سنة | 15 | 100 |
| البالغون | 15 | 100 |
| الحمل والرضاعة | 15 | 100 |
| * الحصة الكافية، لم تحددها RDA/RDI بعد | ||
التحويل : 1 µg = 40 و.د..
المصادر

على الرغم من أن ڤيتامين د ليس موجوداً بشكل طبيعي في معظم الأغذية،[3][5] إلا أنه من الشائع إضافته كمعزز في الأغذية المصنعة. في بعض البلدان، تكون الأغذية الأساسية معززة بڤيتامين د.[27]
المصادر الطبيعية
بصفة عامة، يوجد ڤيتامين د2 في الفطريات ويوجد ڤيتامين د3 في الأغذية الحيوانية.[28][29] ينتج ڤيتامين D2 بتعرض الإرگوسترول، الموجود في الكثير من الفطريات، للأشعة الفوق بنفسجية. تزيد نسبة ڤيتامين د 2 في الفطريات وأشنة الأيل، بزيادة التعرض للأشعة الفوق بنفسجية.[30][31] يتم محاكاة هذه العملية بواسطة مصابيح الأشعة فوق البنفسجية الاصطناعية، حيث تركز مستويات ڤيتامين د 2 على مستويات أعلى.[29]
| المصدر | μg/g | IU/g | |
|---|---|---|---|
| C. arbuscula (أشنة)، الأغصان، مجففة[30] | ڤيتامين د3 | 0.67–2.04 | 27–82 |
| ڤيتامين د2 | 0.22–0.55 | 8.8–22 | |
| الفطر الأبيض (الفطر الشائع): د2 + D3 | |||
| الپورتوبلو | نيء | 0.003 | 0.1 |
| معرض للأشعة الفوق بنفسجية | 0.112 | 4.46 | |
| الكريميني | نيء | 0.001 | 0.03 |
| معرض للأشعة الفوق بنفسجية | 0.319 | 12.76 | |
| المصادر | IU/g | الغير قياسي |
|---|---|---|
| مح البيض المطهو | 0.7 | 44 IU للبيضة وزن 61 گ |
| الكبد البقري المطهو، المطهو ببطيء | 0.5 | |
| زيت كبد السمك، مثل زيد كبد القد | 100 | 450 IU لكل ملعقة صغيرة (4.5 گ) |
| الأسماك الدهنية | ||
| السلمون، النيء، المطهو، المجفف | 5.2 | |
| المكرل، المطهو، المجفف | 4.6 | |
| التونة، المحفوظة في الزيت | 2.7 | |
| السردين، المحفوظ في الزيت، المصفى | 1.9 | |
السبيل التفاعلي
انقر على الأيقونة في الركن الأسفل الأيمن للفتح.
انقر على الجينات والپروتينات والأيضات المبينة أدناه للربط إلى المقالات المتعلقة. [§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "VitaminDSynthesis_WP1531".
الكيمياء الضوئية

التطور
التخليق الصناعي
آلية العمل
التنشيط الاستقلابي

يسري ڤيتامين د في الدم لينتقل إلى الكبد، حيث يتحول إلى الهروم الطليعي، الكالسيفديول. في الكلى يتحول الكالسيفدريول إلى كالسيتريول، الشكل النشط حيوياً من ڤيتامين د.[33]
سواء تم تصنيعه في الجلد أو تناوله، فإن يقوم الكبد بهدركسلة ڤيتامين د عند الموضع 25 (أعلى يمين الجزيء) ليشكل 25 هيدروكسيكولـِكالسيفـِرول (كاليسيفديول أو 25(OH)D).[34] يُحفز هذا التفاعل بواسطة إنزيم مكروي يسمى ڤيتامين د 25-هيدروكسيليز، منتُج الجين البشري CYP2R1، الذي يعبر عنه بواسطة الخلايا الكبدية.[35] بمجرد تخليقه، يُطلق المنتج في الپلازما، حيث يرتبط بپروتين حامل α-گلوبولين يسمى الپروتين المرتبط بڤيتامين د.[36]
يُنقل الكالسيفديول إلى الأنابي القربية للكلى، حيث يُهدركسل عند الموضع 1-α (أسفل يمين الجزيء) ليشكل الكالسيتريول (1,25-dihydroxycholecalciferol, 1,25(OH)2D). يُحفز تحويل الكالسيفديول إلى كالسيتريول عن طريق انزيم 25-hydroxyvitamin D3 1-alpha-hydroxylase، الذي ينتجه الجين البشري CYP27B1. يزداد نشاط الجين CYP27B1 عن طريق الهرمونات الجار درقية، وبانخفاض الكالسيوم أو الفوسفات.[5][33]
بعد خطوة التحويل النهائية في الكلى، يتم تحرير الكالسيتريول في الدورة الدموية. من خلال الارتباط بالپروتين المرتبط بڤيتامين د، ينتقل الكالسيتريول في جميع أنحاء الجسم، بما في ذلك الأعضاء المستهدفة التقليدية مثل الأمعاء والكلى والعظام.[17] الكالسيتريول هو أقوى ربيطعة طبيعية من مستقبل ڤيتامين د، والذي يتوسط معظم العمليات الفسيولوجية لڤيتامين د.[5][33]
بالإضافة إلى الكلى، يُخلق الكالسيتريول أيضاً بواسطة بعض الخلايا الأخرى ومنها [خلية دم بيضاء|خلايا الدم البيضاء]]-وحيدة الخلية في الجهاز المناعي. عندما يتم تخليقه بواسطة خلايا الدم البيضاء-وحيدة الخلية، يعمل الكالسيتريول موضعياً كسيتوكين، معدلاً دفاعات الجسم ضد الجراثيم الغازية عن طريق تحفيز جهاز المناعة الفطري.[33]
التعطيل
قد ينخفض نشاط الكالسيفديول والكالسيتريول عن طريق الهدركسلة عند الموضع 24 بواسطة هيدروكسيليز-24 ڤيتامين د 3، مشكلاً على التوالي، السـِكالسيفرول والكالسيترول.[34]
الاختلاف بين الركائز
يتشارك ڤيتامين د2 (الإرگوكالسيفـِرول) وڤيتامين د3 (الكولساليفرول) آلية عمل متشابهة كما هو موضح أعلاه.[34] المستقبلات المنتجة بواسطة ڤيتامين د2 تسمى أحياناً بالسابقة "إر" أو "إرگو" لتفرقتها عن نظيراتها المنتجة من ڤيتامين د3.[37]
- تميل المستقبلات المنتجة من ڤيتامين د2 للارتباط بشكل أقل مع الپروتين المرتبط بڤيتامين د.
- قد يتهدركسل ڤيتامين د3 ليتحول إلى كالسيفدريول، بواسطة سترول 27-هيدروكسيليز (CYP27A1)، لكن ڤيتامين د2 لا يمكنه ذلك.
- قد يتهدركسل الإرگوكالسيفـِرول مباشرة عن الموضع 24. تؤدي هذه الهدركسلة أيضاً إلى درجة تعطيل أكبر: بينما ينخفض نشاط الكالسيتريول إلى 60% عن النسبة الأصلية بعد 24-هدركسلة،[38] بينما ينخض نشاط الإركالسيتريول بنسبة 10 مرات مقارنة بالإركالسيتترول.[39]
التاريخ
عام 1914 اكتشف الباحثان الأمريكييان إلمر مكولم ومارگريت ديڤيز[11] مادة في زيت كبد القد التي سُميت لاحقاً ڤتامين أ. لاحظ الطبيب البريطاني إدوارد ملانبي أن الكلاب التي كانت تتغذى على زيت كبد القد لم يتطور لديها الرخد (لين العظام)، وخلص إلى أن ڤيتامين أ، أو أي عامل مرتبط به بشكل وثيق، يمكنه الوقاية من هذا المرض. عام 1922، اختبر إلمر مكولم زيت كبد القد المعدل الذي تم بتدمير ڤيتامين أ الموجود فيه.[11] شفيت الكلاب المعالجة بهذا الزيت المعدل، فخلص مكولم إلى أن العامل الموجود في زيت كبد القد الذي يمكنه علاج الرخد يختلف عن ڤيتامين أ. أطلق على هذه المادة اسم ڤيتامين د لأنها كانت رابع ڤيتامين يتم تسميته.[40][41][42] لم يُدرك في البداية أنه على عكس الڤيتامينات الأخرى، يمكن تصنيع ڤيتامين د من قبل البشر من خلال التعرض للأشعة فوق البنفسجية.
عام 1925،[11] ثبت أنه عندما يتم تشعيع 7-ديهيدروكولسترول بالضوء، يتم إنتاج شكل من ڤيتامين قابل للذوبان في الدهون (المعروف الآن باسم ڤيتامين د3). أعلن ألفرد فابيان هس أن: "الضوء يكافيء ڤيتامين د".[43] حصل أدولف ڤينداوس، في جامعة گوتنگن الألمانية، على جائزة نوبل في الكيمياء عام 1928 عن عمله على تركيب على الستيرولات وعلاقتها بالڤيتامينات.[44] عام 1929، كان هناك مجموعة باحثين في المعهد الوطني للأبحاث الطبية في هامپستيد بلندن، تعمل على تركيب ڤيتامين د، والتي لا تزال غير معروفة، فضلاً عن بنية السترويدات. عُقدت اجتماع بين ج.ب.س. هالدان، د.د. برنال، ودروثي كراوفوت، لمناقشة التركيبات المحتملة، والذي أسهم في اجتماع المجموعة معاً. أظهر المسح البلوري بالأشعة السينية أن جزيئات الستيرول كانت مسطحة، وليس كما اقترح الفريق الألماني بقيادة ڤينداوس. عام 1932، نشر أوتو روزنهايم وهارولد كينگ ورقة بحثية طرحا فيها بُنى للستيرولات والأحماض الصفراوية التي وجدت قبولًا فورياً.[45] التعاون الأكاديمي الغير رسمي بين أعضاء الفريق؛ روبرت بندكت بورديلون، أوتو روزنهايم، هارولد كنگ، وكنيث كالو، كان مثمراً للغاية وأدى إلى عزل ومعرفة خصائص ڤيتامين د.[46] في ذلك الوقت، كانت سياسة [[مجلس الأبحاث الطبية (الولايات المتحدة)|مجلس الأبحاث الطبيةي] لا تعطي براءات اختراع للاكتشافات، معتقدة أن نتائج البحث الطبي ينبغي أن تكون متاحة للجميع. في الثلاثينيات، توصل ڤينداوس للمزيد من المعلومات حول التركيب الكيميائي لڤيتامين د.[47]
عام 1923، أثبت الكيميائي الحيوي الأمريكي هاري ستينبوك في جامعة وسكنسن أن التشعيش بالضوء الفوق بنفسجي يزيد من محتوى ڤيتامين د في الأغذية والمواد العضوية الأخرى.[48] بعد تشعيش غذاء الجرذان، اكتشف ستينبوك أن الجرذان قد شفيت من الرخد. وعُرف أن نقص ڤيتامين د يسبب الإصابة بالرخد. مستخدماً 300 دولاراً من ماله الخاص، حصل ستينبوك على براءة لاتشافه. استخدمت تقنيته التشعيعية للأغذية المعلبة، وأشهرها الحليب. بانتهاء براءة اختراعه عام 1945، تم القضاء على الرخد تقريباً في الولايات المتحدة.[49]
عام 1969، بعد دراسة الأجزاء النووية لخلايا الأمعاء، تمكن مارك هوسلر وتوني نورمان من تحديد پروتين محدد لڤيتامين د يسمى مستقبل ڤيتامين د.[50] عام 1971-72، أُكتشف أن هناك المزيد من عمليات الأيض لڤيتامين د للأشكال النشطة منه. في الكبد، وُجد أن ڤيتامين د يتحول إلى كالسيفديول. بعد ذلك يتحول الكالسيفديول في الكبد إلى كالسيتريول، الشكل النشط حيوياً من ڤيتامين د.[10] يسري الكالسيتريول في الدم كهرمون، حيث ينشط تركيز الكالسيوم والفوسفات في الدورة الدموية ويعزز النمو الصحي ويعيد تشكيل العظام. مستقلبات ڤيتامين د، الكالسيفديول والكالسيتريول، تم التعرف عليهما بواسطة فريقين متنافسين، أحدمها بقيادة مايكل هوليك في معمل هكتور دلوسا وبواسطة توني نورمان وزملائه.[51][52][53]
الأبحاث
هناك نشاط بحثي كبير حول تأثيرات ڤيتامين د ومستقلباته في النماذج الحيوانية وأنظمة الخلايا ودراسات التعبير الجيني وعلم الأوبئة والعلاجات السريرية. قد تنتج هذه الأنواع المختلفة من الدراسات أدلة متضاربة حول فوائد التدخلات مع ڤيتامين د.[54] تؤكد إحدى المدارس الفكرية أن فسيولوجيا الإنسان مضبوطة بدقة لتتوافق مع تناول 4000-12000 وحدة دولية/يوميًا من التعرض لأشعة الشمس مع ما يصاحب ذلك من مصل 25-هيدروكسي ڤيتامين د من 40 إلى 80 ن.ج/مل[55] وهذا هو المطلوب من أجل الحياة الصحية المثلى. مؤيدو هذا الرأي، ومن بينهم بعض أعضاء اللجنة التي صاغت تقرير عام 1997 الذي تم استبداله الآن بشأن ڤيتامين د من المنظمة الدولية للهجرة، يؤكدون أن تحذير معهد الطب الأمريكي بشأن تركيزات المصل فوق 50 ن.ج./مل يفتقر إلى المعقولية الحيوية. ويقترحون، بالنسبة لبعض الأشخاص، أن الحد من مخاطر الأمراض التي يمكن الوقاية منها يتطلب مستوى أعلى من ڤيتامين د من ذلك الذي أوصى به معهد الطب الأمريكي.[55][56] أطلق مكتب المكملات الغذائية التابع للمعاهد الوطنية للصحة بالولايات المتحدة مبادرة ڤيتامين د في عام 2014 لتتبع الأبحاث الحالية وتوفير التعليم للمستهلكين.[57] في تنقيح عام 2016، أدركوا أن مجموعة متزايدة من الأبحاث تشير إلى أن ڤيتامين د قد يلعب دورًا ما في الوقاية والعلاج من مرض السكري من النمط الأول والثاني، وعدم تحمل الگلوكوز، وارتفاع ضغط الدم، والتصلب المتعدد، والحالات الطبية الأخرى. ويذكرون كذلك: "ومع ذلك، فإن معظم الأدلة على هذه الأدوار تأتي من الدراسات المختبرية والحيوانية والوبائية، وليس من التجارب السريرية العشوائية التي تعتبر أكثر تحديدًا. وإلى أن يتم إجراء مثل هذه التجارب، فإن آثار الأدلة المتاحة على الصحة العامة وسيتم مناقشة رعاية المرضى".[6]
تربط بعض الدراسات الأولية بين مستويات ڤيتامين د المنخفضة والمرض في مراحل عمرية لاحقة.[58] اعتباراً من 2013، كانت الأدلة غير كافية لتحديد ما إذا كان ڤيتامين د يؤثر على خطر الإصابة بالسرطان.[59] وجد أحد التحاليل التلوية انخفاضاً في معدل الوفيات لدى كبار السن.[14] خلص تحليل تلوي آخر شمل أكثر من 350.000 شخص إلى أن مكملات ڤيتامين د للأفراد الذين يعيشون في المجتمعات الغير مميزة لا تقلل من النتائج الهيكلية (الكسر الكلي) أو النتائج غير الهيكلية (احتشاء عضلة القلب، وأمراض القلب الإقفارية، والسكتة الدماغية، وأمراض الأوعية الدموية الدماغية، والسرطان) بأكثر من 15%، وأن التجارب البحثية الإضافية ذات التصميم المماثل من غير المرجح أن تغير هذه الاستنتاجات.[15] وجد التحليل التلوي لعام 2019 أنه قد يكون هناك خطر متزايد للإصابة بالسكتة الدماغية عند تناول كل من الكالسيوم وڤيتامين د.[60] يمكن أن يعزى دور ڤيتامين د في تكلس الأوعية الدموية إلى هذه النتائج.[61]
ينتشر نقص ڤيتامين د بين سكان أوروپا.[62] تقوم الأبحاث الأوروپية بتقييم مستويات تناول ڤيتامين د بالاقتران مع معدلات المرض وسياسات التوصيات الغذائية، وإغناء الطعام، ومكملات ڤيتامين د، وكميات صغيرة من التعرض لأشعة الشمس.[63]
بصرف النظر عن تنشيط VDR، هناك العديد من الآليات البديلة للعمل قيد الدراسة، مثل تثبيط تحويل الإشارة عن طريق القنفذية، وهو هرمون يشارك في التخلق.[64]
المصادر
- ^ MacDonald, James (18 July 2019). "How Does the Body Make Vitamin D from Sunlight?". JSTOR Daily. Retrieved 22 July 2019.
- ^ Holick MF, MacLaughlin JA, Clark MB, Holick SA, Potts JT, Anderson RR, et al. (10 October 1980). "Photosynthesis of previtamin D3 in human skin and the physiologic consequences". Science. 210 (4466): 203–5. Bibcode:1980Sci...210..203H. doi:10.1126/science.6251551. ISSN 0036-8075. JSTOR 1685024. PMID 6251551.
- ^ أ ب خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةHolick_2006 - ^ Calvo MS, Whiting SJ, Barton CN (February 2005). "Vitamin D intake: a global perspective of current status". The Journal of Nutrition. 135 (2): 310–6. doi:10.1093/jn/135.2.310. PMID 15671233.
- ^ أ ب ت ث ج ح Norman AW (August 2008). "From vitamin D to hormone D: fundamentals of the vitamin D endocrine system essential for good health". The American Journal of Clinical Nutrition. 88 (2): 491S–9S. doi:10.1093/ajcn/88.2.491S. PMID 18689389.
- ^ أ ب ت ث ج "Vitamin D Fact Sheet for Health Professionals". National Institutes of Health (NIH). February 11, 2016. Retrieved June 6, 2017.
 هذا المقال يضم نصاً من هذا المصدر، الذي هو مشاع.
هذا المقال يضم نصاً من هذا المصدر، الذي هو مشاع.
- ^ Dierkes, J; Stangl, GI; Pot, GK; Erkkilä, AT; Hansen, AL; Lindqvist, H; Lauritzen, L; Mellgren, G; Ueland, PM; Gudbrandsen, OA; Mueller-Belecke, A; Hirche, F; Gjessing, HR; Lehmann, U (October 2015). "Efficacy of fish intake on vitamin D status: a meta-analysis of randomized controlled trials". The American Journal of Clinical Nutrition. 102 (4): 837–847. doi:10.3945/ajcn.114.105395. PMID 26354531.
- ^ "Vitamin D Tests". Lab Tests Online (USA). American Association for Clinical Chemistry. Retrieved June 23, 2013.
- ^ Hollis BW (January 1996). "Assessment of vitamin D nutritional and hormonal status: what to measure and how to do it". Calcified Tissue International. 58 (1): 4–5. doi:10.1007/BF02509538. PMID 8825231.
- ^ أ ب Holick MF, Schnoes HK, DeLuca HF, Suda T, Cousins RJ (July 1971). "Isolation and identification of 1,25-dihydroxycholecalciferol. A metabolite of vitamin D active in intestine". Biochemistry. 10 (14): 2799–804. doi:10.1021/bi00790a023. PMID 4326883.
- ^ أ ب ت ث Wolf G (June 2004). "The discovery of vitamin D: the contribution of Adolf Windaus". The Journal of Nutrition. 134 (6): 1299–302. doi:10.1093/jn/134.6.1299. PMID 15173387.
- ^ Pittas AG, Chung M, Trikalinos T, Mitri J, Brendel M, Patel K, et al. (March 2010). "Systematic review: Vitamin D and cardiometabolic outcomes". Annals of Internal Medicine. 152 (5): 307–14. doi:10.7326/0003-4819-152-5-201003020-00009. PMC 3211092. PMID 20194237.
- ^ خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةBalk09 - ^ أ ب خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةBj2014 - ^ أ ب خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةFutil2014 - ^ Dorland's Illustrated Medical Dictionary, under Vitamin (Table of Vitamins)
- ^ أ ب ت "About Vitamin D". University of California, Riverside. November 2011. Retrieved January 24, 2015.
- ^ Advanced Dairy Chemistry, Volume 2: Lipids, Second Edition p. 377
- ^ بي بي سي العربية
- ^ كنانة أونلاين
- ^ "Vitamin D supplementation to the older adult population in Germany has the cost‐saving potential of preventing almost 30,000 cancer deaths per year". onlinelibrary.wiley.com. 2021-02-04. Retrieved 2021-02-10.
- ^ أ ب خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةRoss_2011 - ^ خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةHCanada - ^ خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةAusNZ - ^ خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةEFSA-vitD - ^ خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةEFSA-2012 - ^ Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes (1997). DRI, Dietary reference intakes: for calcium, phosphorus, magnesium, vitamin D, and fluoride. Washington, D.C: National Academy Press. p. 250. doi:10.17226/5776. ISBN 978-0-309-06350-0. PMID 23115811.
- ^ خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةpmid1297827 - ^ أ ب Keegan RJ, Lu Z, Bogusz JM, Williams JE, Holick MF (January 2013). "Photobiology of vitamin D in mushrooms and its bioavailability in humans". Dermato-Endocrinology. 5 (1): 165–76. doi:10.4161/derm.23321. PMC 3897585. PMID 24494050.
- ^ أ ب Wang T, Bengtsson G, Kärnefelt I, Björn LO (September 2001). "Provitamins and vitamins D₂ and D₃ in Cladina spp. over a latitudinal gradient: possible correlation with UV levels". Journal of Photochemistry and Photobiology. B, Biology (Submitted manuscript). 62 (1–2): 118–22. doi:10.1016/S1011-1344(01)00160-9. PMID 11693362.
- ^ Haytowitz DB (2009). "Vitamin D in mushrooms" (PDF). Nutrient Data Laboratory, US Department of Agriculture. Retrieved April 16, 2018.
 هذا المقال يضم نصاً من هذا المصدر، الذي هو مشاع.
هذا المقال يضم نصاً من هذا المصدر، الذي هو مشاع.
- ^ "Search, National Nutrient Database for Standard Reference Release 27". US Department of Agriculture, Agricultural Research Service. 2014. Retrieved June 12, 2015.
 هذا المقال يضم نصاً من هذا المصدر، الذي هو مشاع.
هذا المقال يضم نصاً من هذا المصدر، الذي هو مشاع.
- ^ أ ب ت ث Adams JS, Hewison M (February 2010). "Update in vitamin D". The Journal of Clinical Endocrinology and Metabolism. 95 (2): 471–8. doi:10.1210/jc.2009-1773. PMC 2840860. PMID 20133466.
- ^ أ ب ت Bikle, DD (March 20, 2014). "Vitamin D metabolism, mechanism of action, and clinical applications". Chemistry & Biology. 21 (3): 319–29. doi:10.1016/j.chembiol.2013.12.016. PMC 3968073. PMID 24529992.
{{cite journal}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Cheng JB, Levine MA, Bell NH, Mangelsdorf DJ, Russell DW (May 2004). "Genetic evidence that the human CYP2R1 enzyme is a key vitamin D 25-hydroxylase". Proceedings of the National Academy of Sciences of the United States of America. 101 (20): 7711–5. Bibcode:2004PNAS..101.7711C. doi:10.1073/pnas.0402490101. PMC 419671. PMID 15128933.
- ^ Laing CJ, Cooke NE (2004). "Section I: Ch. 8: Vitamin D Binding Protein". In Feldman D, Glorieux FH, Pike JW (eds.). Vitamin D. Vol. 1 (2 ed.). Academic Press. pp. 117–134. ISBN 978-0122526879.
- ^ "IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN): Nomenclature of vitamin D. Recommendations 1981". European Journal of Biochemistry. 124 (2): 223–7. May 17, 1982. doi:10.1111/j.1432-1033.1982.tb06581.x. PMID 7094913.
- ^ Holick MF, Kleiner-Bossaller A, Schnoes HK, Kasten PM, Boyle IT, DeLuca HF (October 1973). "1,24,25-Trihydroxyvitamin D3. A metabolite of vitamin D3 effective on intestine". The Journal of Biological Chemistry. 248 (19): 6691–6. PMID 4355503.
- ^ Horst RL, Reinhardt TA, Ramberg CF, Koszewski NJ, Napoli JL (July 1986). "24-Hydroxylation of 1,25-dihydroxyergocalciferol. An unambiguous deactivation process". The Journal of Biological Chemistry. 261 (20): 9250–6. PMID 3013880.
- ^ Carere, Suzanne (يوليو 25, 2007). "Age-old children's disease back in force". Toronto Star. Archived from the original on مايو 17, 2008. Retrieved أغسطس 24, 2010.
{{cite news}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Conis, Elena (July 24, 2006). "Fortified foods took out rickets". Los Angeles Times. Retrieved August 24, 2010.
{{cite news}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ McClean FC, Budy AM (January 28, 1964). "Vitamin A, Vitamin D, Cartilage, Bones, and Teeth". Vitamins and Hormones. Vol. 21. Academic Press. pp. 51–52. ISBN 978-0-12-709821-0.
{{cite book}}: External link in|chapterurl=|chapterurl=ignored (|chapter-url=suggested) (help) - ^ "History of Vitamin D". University of California at Riverside. 2011. Retrieved May 9, 2014.
- ^ "Adolf Windaus – Biography". Nobelprize.org. March 25, 2010. Retrieved March 25, 2010.
- ^ Rosenheim O, King H (1932). "The Ring-system of sterols and bile acids. Part II". J. Chem. Technol. Biotechnol. 51 (47): 954–7. doi:10.1002/jctb.5000514702.
- ^ Askew FA, Bourdillon RB, Bruce HM, Callow RK, St. L. Philpot J, Webster TA (1932). "Crystalline Vitamin D". Proceedings of the Royal Society of London. Series B, Containing Papers of a Biological Character. 109 (764): 488–506. doi:10.1098/rspb.1932.0008. JSTOR 81571.
- ^ Hirsch AL (2011). "Industrial aspects of vitamin D". In Feldman DJ, Pike JW, Adams JS (eds.). Vitamin D. Academic Press. p. 73. ISBN 978-0-12-387035-3.
- ^ Ziedonis AA, Mowery DC, Nelson RR, Bhaven NS (2004). Ivory tower and industrial innovation: university-industry technology transfer before and after the Bayh-Dole Act in the United States. Stanford Business Books. pp. 39–40. ISBN 978-0-8047-4920-6.
- ^ Marshall, James (September 2010). Elbridge a Stuart: Founder of Carnation Company. Kessinger Publishing. ISBN 978-1-164-49678-6.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Haussler MR, Norman AW (January 1969). "Chromosomal receptor for a vitamin D metabolite". Proceedings of the National Academy of Sciences of the United States of America. 62 (1): 155–62. Bibcode:1969PNAS...62..155H. doi:10.1073/pnas.62.1.155. PMC 285968. PMID 5253652.
- ^ Holick MF, Schnoes HK, DeLuca HF (April 1971). "Identification of 1,25-dihydroxycholecalciferol, a form of vitamin D3 metabolically active in the intestine". Proceedings of the National Academy of Sciences of the United States of America. 68 (4): 803–4. Bibcode:1971PNAS...68..803H. doi:10.1073/pnas.68.4.803. PMC 389047. PMID 4323790.
- ^ Norman AW, Myrtle JF, Midgett RJ, Nowicki HG, Williams V, Popják G (July 1971). "1,25-dihydroxycholecalciferol: identification of the proposed active form of vitamin D3 in the intestine". Science. 173 (3991): 51–4. Bibcode:1971Sci...173...51N. doi:10.1126/science.173.3991.51. PMID 4325863.
- ^ Holick MF, DeLuca HF, Avioli LV (January 1972). "Isolation and identification of 25-hydroxycholecalciferol from human plasma". Archives of Internal Medicine. 129 (1): 56–61. doi:10.1001/archinte.1972.00320010060005. PMID 4332591.
- ^ Dankers W, Colin EM, van Hamburg JP, Lubberts E (2016). "Vitamin D in Autoimmunity: Molecular Mechanisms and Therapeutic Potential". Front Immunol. 7: 697. doi:10.3389/fimmu.2016.00697. PMC 5247472. PMID 28163705.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ أ ب Heaney RP, Holick MF (March 2011). "Why the IOM recommendations for vitamin D are deficient". Journal of Bone and Mineral Research. 26 (3): 455–7. doi:10.1002/jbmr.328. PMID 21337617.
- ^ Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, et al. (July 2011). "Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline". The Journal of Clinical Endocrinology and Metabolism. 96 (7): 1911–30. doi:10.1210/jc.2011-0385. PMID 21646368.
- ^ "ODS Vitamin D Initiative". Office of Dietary Supplements, US National Institutes of Health. 2014.
- ^ Pyrżak B, Witkowska-Sędek E, Krajewska M, Demkow U, Kucharska AM (2015). "Metabolic and immunological consequences of vitamin D deficiency in obese children". Body Metabolism and Exercise. Advances in Experimental Medicine and Biology. Vol. 840. pp. 13–9. doi:10.1007/5584_2014_81. ISBN 978-3-319-10249-8. PMID 25315624.
- ^ "How is vitamin D being studied now in clinical cancer research?". National Cancer Institute, US National Institutes of Health. October 21, 2013.
- ^ Khan SU, Khan MU, Riaz H, Valavoor S, Zhao D, Vaughan L, et al. (July 9, 2019). "Effects of Nutritional Supplements and Dietary Interventions on Cardiovascular Outcomes". Annals of Internal Medicine. 171 (3): 190–198. doi:10.7326/m19-0341. ISSN 0003-4819. PMID 31284304.
- ^ Wang, Jeffrey; Zhou, Jimmy; Robertson, Graham; Lee, Vincent (2018-05-22). "Vitamin D in Vascular Calcification: A Double-Edged Sword?". Nutrients (in الإنجليزية). 10 (5): 652. doi:10.3390/nu10050652. ISSN 2072-6643. PMC 5986531. PMID 29786640.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Cashman KD, Dowling KG, Škrabáková Z, Gonzalez-Gross M, Valtueña J, De Henauw S, et al. (April 2016). "Vitamin D deficiency in Europe: pandemic?". The American Journal of Clinical Nutrition. 103 (4): 1033–44. doi:10.3945/ajcn.115.120873. PMC 5527850. PMID 26864360.
- ^ خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةspiro - ^ Sarkar FH, Li Y, Wang Z, Kong D (September 2010). "The role of nutraceuticals in the regulation of Wnt and Hedgehog signaling in cancer". Cancer Metastasis Reviews. 29 (3): 383–94. doi:10.1007/s10555-010-9233-4. PMC 2974632. PMID 20711635.
قراءات إضافية
وصلات خارجية
- "Vitamin D". Drug Information Portal. U.S. National Library of Medicine.
- "Ergocalciferol". Drug Information Portal. U.S. National Library of Medicine.
- "Cholecalciferol". Drug Information Portal. U.S. National Library of Medicine.
- Vitamin D in children from the Royal National Orthopaedic Hospital Trust