تفاعل الألدول
(تم التحويل من Aldol reaction)
تفاعل الألدول هو aldol reaction وسيلة لتشكيل روابط الكربون-كربون في الكمياء العضوية.[1][2][3] اكتشفه بشكل مستقل تشارلز-أدولف ڤورتز[4][5][6] وألكسندر بورودين عام 1872،[7]

The flask on the right is a solution of lithium diisopropylamide (LDA) in tetrahydrofuran (THF). The flask on the left is a solution of the lithium enolate of tert-butyl propionate (formed by addition of LDA to tert-butyl propionate). An aldehyde can then be added to the enolate flask to initiate an aldol addition reaction.
Both flasks are submerged in a dry ice/acetone cooling bath (−78 °C) the temperature of which is being monitored by a thermocouple (the wire on the left).
الآليات
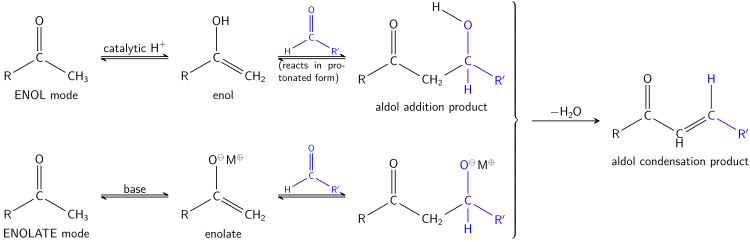
آلية الإنول

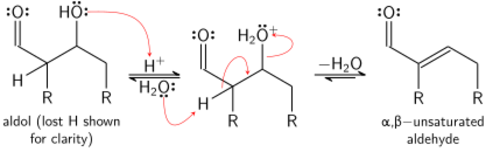
آلية الإنولات


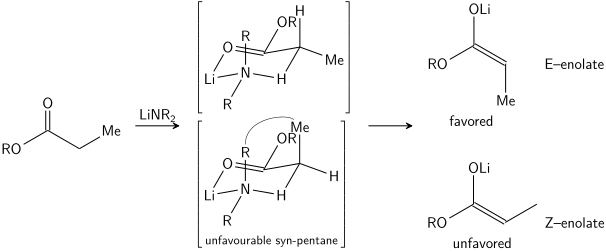
نموذج زيمرمان-تريكسلر
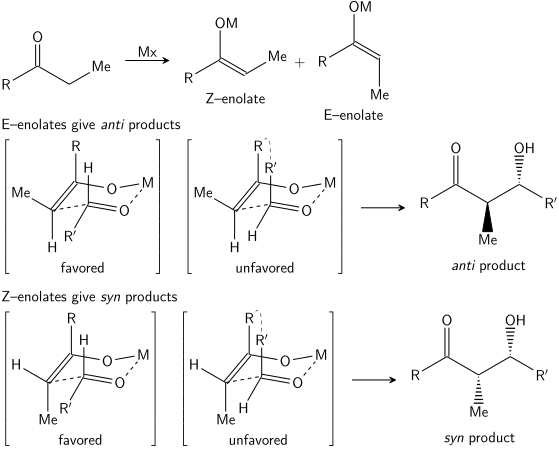
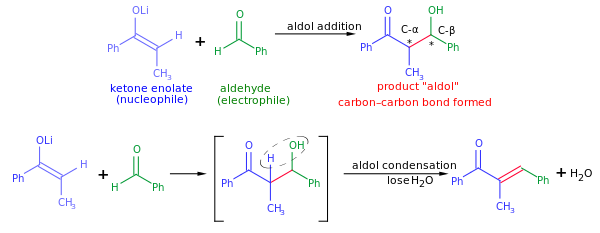
السيطرة المتفاعلة عبر الأدول
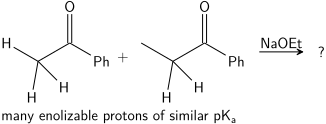
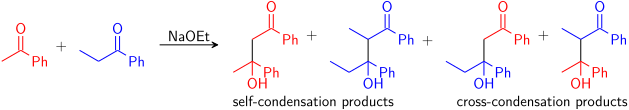
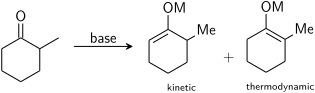
الحموضة
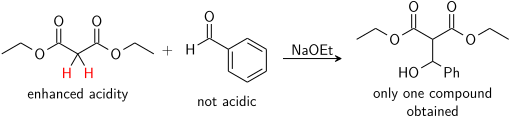
ترتيب الإضافة
الإنولات
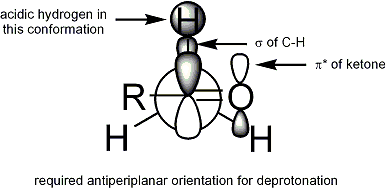
الهندسة
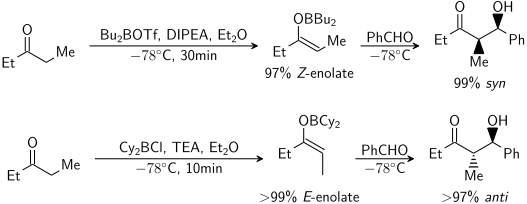
الكيمياء الموضعية
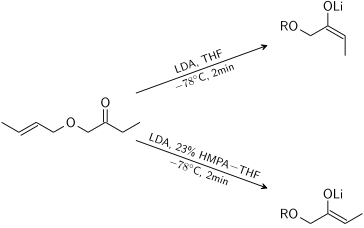
Stereoselectivity
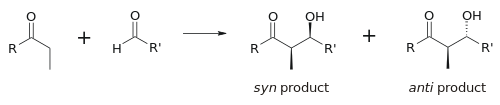
هندسة الإنوليت
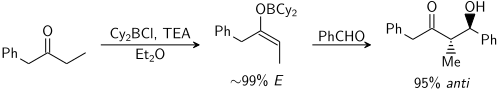
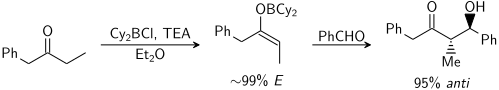
أيو الفلز
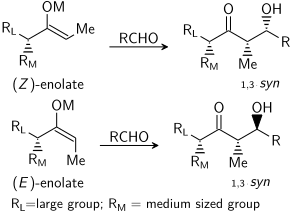
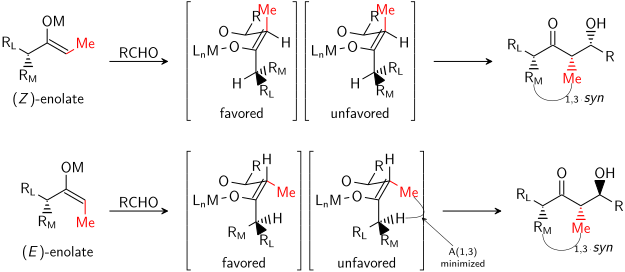
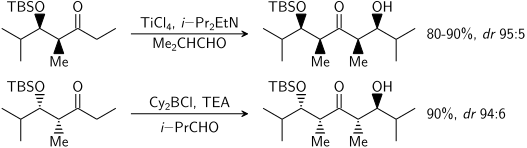
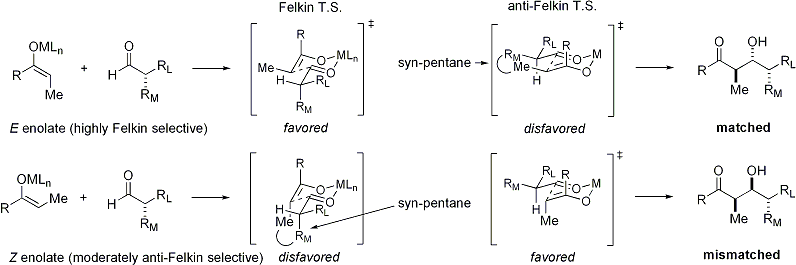
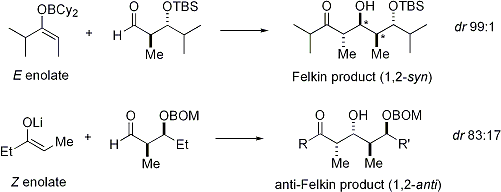
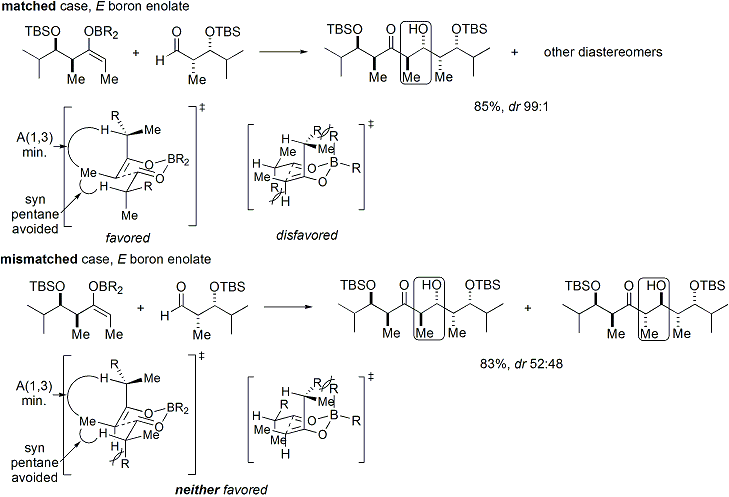
=كمياء أوكسزوليدينون إيفانز
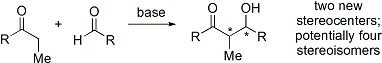
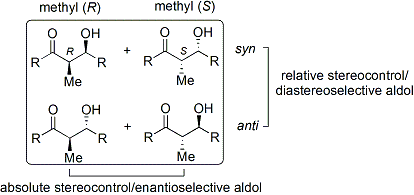
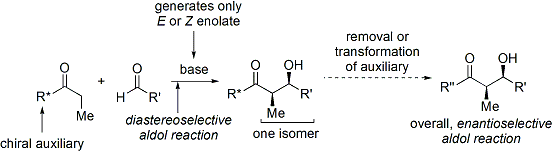
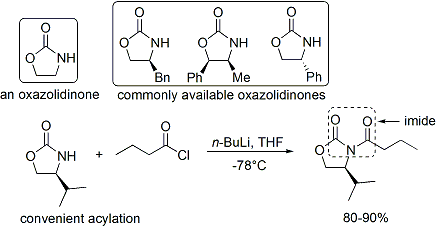
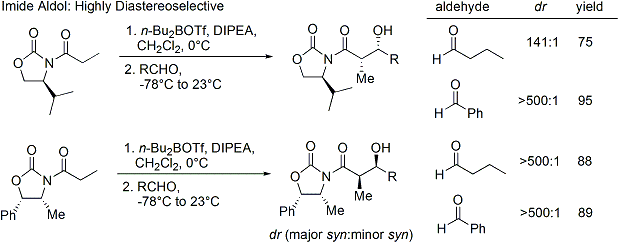
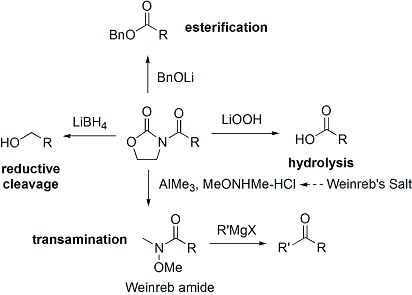
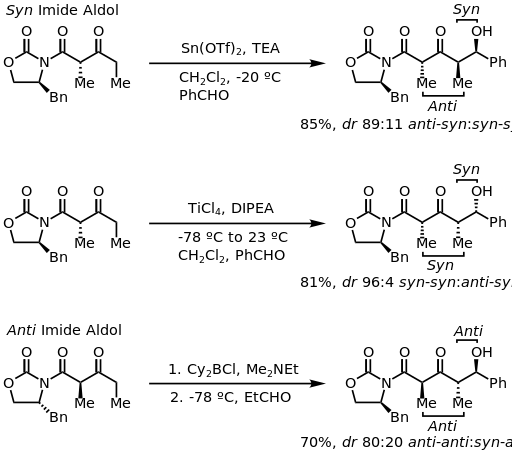
الاختفالات والطرق الحديثة
تفاعلات خلات الأدول
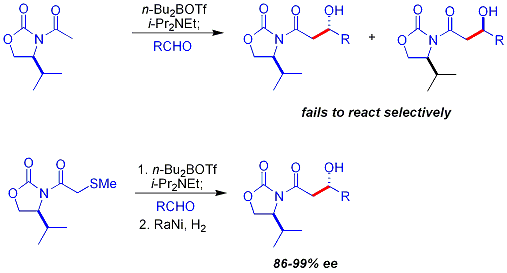
تفاعل أدول موكايايما

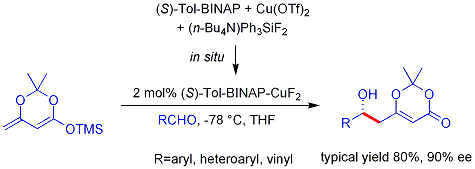
Crimmins thiazolidinethione aldol
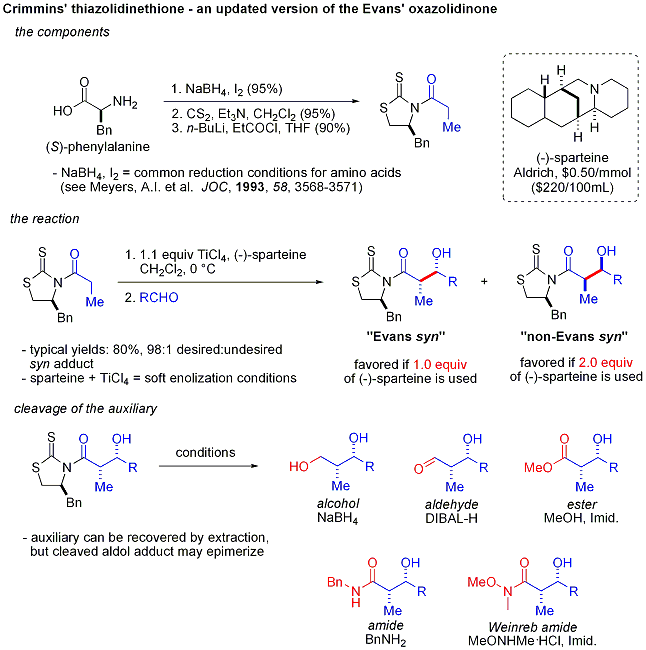
Organocatalysis
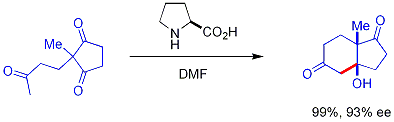
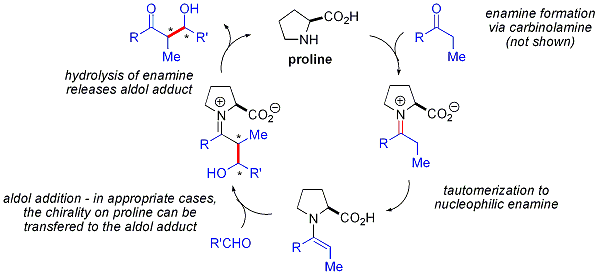
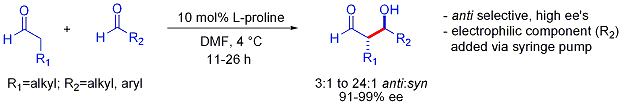
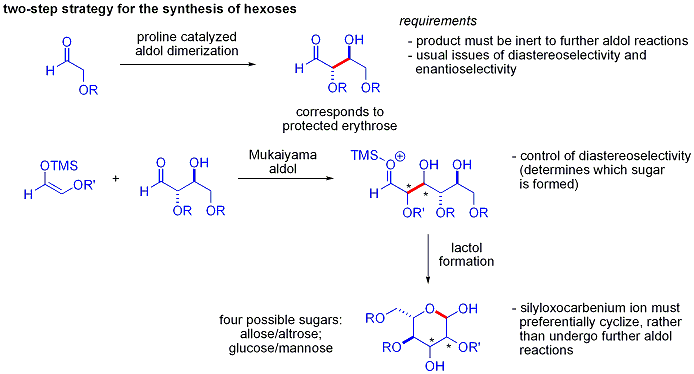
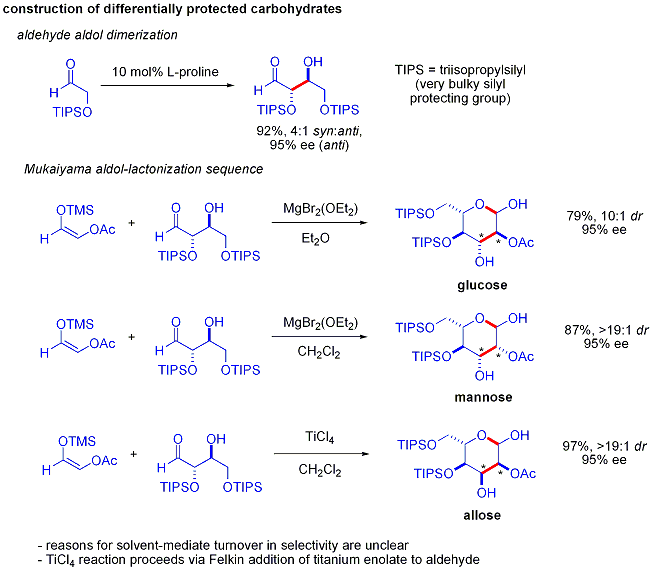
إضافات الأدول "المباشرة"
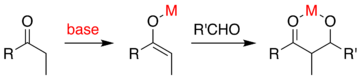
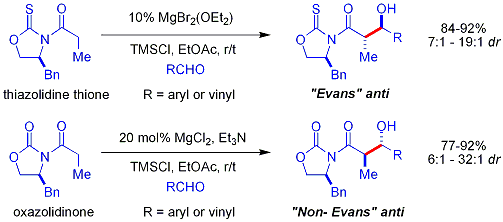
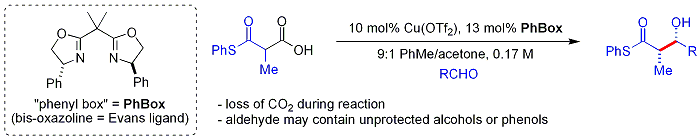
تفاعلات الأدول البيولوجية
انظر أيضاً
المصادر
- ^ Wade, L. G. (2005). Organic Chemistry (6th ed.). Upper Saddle River, New Jersey: Prentice Hall. pp. 1056–66. ISBN 0-13-236731-9.
- ^ Smith, M. B.; March, J. (2001). Advanced Organic Chemistry (5th ed.). New York: Wiley Interscience. pp. 1218–23. ISBN 0-471-58589-0.
- ^ Mahrwald, R. (2004). Modern Aldol Reactions, Volumes 1 and 2. Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA. pp. 1218–23. ISBN 3-527-30714-1.
- ^
Wurtz, C. A. (1872). Bull. Soc. Chim. Fr. 17: 436–442.
{{cite journal}}: CS1 maint: untitled periodical (link) - ^ Wurtz, C. A. (1872). "Ueber einen Aldehyd-Alkohol". Journal für Praktische Chemie. 5 (1): 457–464. doi:10.1002/prac.18720050148.
- ^
Wurtz, C. A. (1872). "Sur un aldéhyde-alcool". Comptes rendus de l'Académie des sciences (in French). 74: 1361.
{{cite journal}}: CS1 maint: unrecognized language (link) - ^ Borodin observed the dimerization of acetaldehyde to 3-hydroxybutanal under acidic conditions