حمض النيتروز
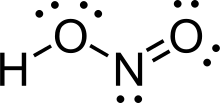
Nitrous acid (molecular formula HNO 2) is a weak and monoprotic acid known only in solution, in the gas phase and in the form of nitrite (NO−2) salts.[2] Nitrous acid is used to make diazonium salts from amines. The resulting diazonium salts are reagents in azo coupling reactions to give azo dyes.
| |
| الأسماء | |
|---|---|
| اسم أيوپاك
Nitrous acid[1]
| |
| Identifiers | |
| رقم CAS | |
3D model (JSmol)
|
|
| 3DMet | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.029.057 |
| رقم EC |
|
| مرجع Gmelin | 983 |
| KEGG | |
| عناوين مواضيع طبية MeSH | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| InChI | InChI={{{value}}} |
| SMILES | |
| الخصائص | |
| الصيغة الجزيئية | HNO2 |
| كتلة مولية | 47.013 g/mol |
| المظهر | Pale blue solution |
| الكثافة | Approx. 1 g/ml |
| نقطة الانصهار | |
| الحموضة (pKa) | 3.15 |
| المخاطر | |
| NFPA 704 (معيـَّن النار) | |
| نقطة الوميض | Non-flammable |
| مركبات ذا علاقة | |
أنيونات أخرى
|
Nitric acid |
كاتيونات أخرى
|
Sodium nitrite Potassium nitrite Ammonium nitrite |
مركـّبات ذات علاقة
|
Dinitrogen trioxide |
ما لم يُذكر غير ذلك، البيانات المعطاة للمواد في حالاتهم العيارية (عند 25 °س [77 °ف]، 100 kPa). | |
| مراجع الجدول | |
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Structure
In the gas phase, the planar nitrous acid molecule can adopt both a syn and an anti form. The anti form predominates at room temperature, and IR measurements indicate it is more stable by around 2.3 kJ/mol.[2]
Dimensions of the anti form
(from the microwave spectrum)Model of the anti form
Preparation
Nitrous acid is usually generated by acidification of aqueous solutions of sodium nitrite with a mineral acid. The acidification is usually conducted at ice temperatures, and the HNO2 is consumed in situ.[3][4] Free nitrous acid is unstable and decomposes rapidly.
Nitrous acid can also be produced by dissolving dinitrogen trioxide in water according to the equation
- N2O3 + H2O → 2 HNO2
Reactions
Nitrous acid is the main chemphore in the Liebermann reagent, used to spot-test for alkaloids.
Decomposition
Gaseous nitrous acid, which is rarely encountered, decomposes into nitrogen dioxide, nitric oxide, and water:
- 2 HNO2 → NO2 + NO + H2O
Nitrogen dioxide disproportionates into nitric acid and nitrous acid in aqueous solution:[5]
- 2 NO2 + H2O → HNO3 + HNO2
In warm or concentrated solutions, the overall reaction amounts to production of nitric acid, water, and nitric oxide:
- 3 HNO2 → HNO3 + 2 NO + H2O
The nitric oxide can subsequently be re-oxidized by air to nitric acid, making the overall reaction:
- 2 HNO2 + O2 → 2 HNO3
Reduction
With I− and Fe2+ ions, NO is formed:[6]
- 2 HNO2 + 2 KI + 2 H2SO4 → I2 + 2 NO + 2 H2O + 2 K2SO4
- 2 HNO2 + 2 FeSO4 + 2 H2SO4 → Fe2(SO4)3 + 2 NO + 2 H2O + K2SO4
With Sn2+ ions, N2O is formed:
- 2 HNO2 + 6 HCl + 2 SnCl2 → 2 SnCl4 + N2O + 3 H2O + 2 KCl
With SO2 gas, NH2OH is formed:
- 2 HNO2 + 6 H2O + 4 SO2 → 3 H2SO4 + K2SO4 + 2 NH2OH
With Zn in alkali solution, NH3 is formed:
- 5 H2O + KNO2 + 3 Zn → NH3 + KOH + 3 Zn(OH)2
With N 2H+5, both HN3 and (subsequently) N2 gas are formed:
- HNO2 + [N2H5]+ → HN3 + H2O + H3O+
- HNO2 + HN3 → N2O + N2 + H2O
Oxidation by nitrous acid has a kinetic control over thermodynamic control, this is best illustrated that dilute nitrous acid is able to oxidize I− to I2, but dilute nitric acid cannot.
- I2 + 2 e− ⇌ 2 I− Eo = +0.54 V
- NO−3 + 3 H+ + 2 e− ⇌ HNO2 + H2O Eo = +0.93 V
- HNO2 + H+ + e− ⇌ NO + H2O Eo = +0.98 V
It can be seen that the values of Eocell for these reactions are similar, but nitric acid is a more powerful oxidizing agent. Base on the fact that dilute nitrous acid can oxidize iodide into iodine, it can be deduced that nitrous is a faster, rather than a more powerful, oxidizing agent than dilute nitric acid.[6]
Organic chemistry
Nitrous acid is used to prepare diazonium salts:
- HNO2 + ArNH2 + H+ → ArN+2 + 2 H2O
where Ar is an aryl group.
Such salts are widely used in organic synthesis, e.g., for the Sandmeyer reaction and in the preparation azo dyes, brightly colored compounds that are the basis of a qualitative test for anilines.[7] Nitrous acid is used to destroy toxic and potentially explosive sodium azide. For most purposes, nitrous acid is usually formed in situ by the action of mineral acid on sodium nitrite:[8] It is mainly blue in colour
- NaNO2 + HCl → HNO2 + NaCl
- 2 NaN3 + 2 HNO2 → 3 N2 + 2 NO + 2 NaOH
Reaction with two α-hydrogen atoms in ketones creates oximes, which may be further oxidized to a carboxylic acid, or reduced to form amines. This process is used in the commercial production of adipic acid.
Nitrous acid reacts rapidly with aliphatic alcohols to produce alkyl nitrites, which are potent vasodilators:
- (CH3)2CHCH2CH2OH + HNO2 → (CH3)2CHCH2CH2ONO + H2O
The carcinogens called nitrosamines are produced, usually not intentionally, by the reaction of nitrous acid with secondary amines:
- HNO2 + R2NH → R2N-NO + H2O
Atmosphere of the Earth
Nitrous acid is involved in the ozone budget of the lower atmosphere, the troposphere. The heterogeneous reaction of nitric oxide (NO) and water produces nitrous acid. When this reaction takes place on the surface of atmospheric aerosols, the product readily photolyses to hydroxyl radicals.[9][10]
See also
المصادر
- ^ https://pubchem.ncbi.nlm.nih.gov/compound/24529#section=IUPAC-Name&fullscreen=true
- ^ أ ب Greenwood, N. N. (1997). Chemistry of the Elements (2nd Edition ed.). Oxford:Butterworth-Heinemann. ISBN 0-7506-3365-4.
{{cite book}}:|edition=has extra text (help); Unknown parameter|coauthors=ignored (|author=suggested) (help) p. 462. - ^ "Ethyl Glycidate from (S)-Serine: Ethyl (R)-(+)-2,3-Epoxypropanoate". Org. Synth. 75: 37. 1998. doi:10.15227/orgsyn.075.0037.
{{cite journal}}: Cite uses deprecated parameter|authors=(help) - ^ "Synthesis of 4-, 5-, and 6-methyl-2,2'-bipyridine by a Negishi Cross-coupling Strategy: 5-methyl-2,2'-bipyridine". Org. Synth. 78: 51. 2002. doi:10.15227/orgsyn.078.0051.
{{cite journal}}: Cite uses deprecated parameter|authors=(help) - ^ Kameoka, Yohji; Pigford, Robert (February 1977). "Absorption of Nitrogen Dioxide into Water, Sulfuric Acid, Sodium Hydroxide, and Alkaline Sodium Sulfite Aqueous". Ind. Eng. Chem. Fundamen. 16 (1): 163–169. doi:10.1021/i160061a031.
- ^ أ ب Catherine E. Housecroft; Alan G. Sharpe (2008). "Chapter 15: The group 15 elements". Inorganic Chemistry, 3rd Edition. Pearson. p. 449. ISBN 978-0-13-175553-6.
- ^ Clarke, H. T.; Kirner, W. R. "Methyl Red" Organic Syntheses, Collected Volume 1, p.374 (1941). "Archived copy" (PDF). Archived from the original (PDF) on 2007-09-30. Retrieved 2007-07-26.
{{cite web}}: CS1 maint: archived copy as title (link) - ^ Prudent practices in the laboratory: handling and disposal of chemicals. Washington, D.C.: National Academy Press. 1995. doi:10.17226/4911. ISBN 978-0-309-05229-0.
- ^ Spataro, F; Ianniello, A (November 2014). "Sources of atmospheric nitrous acid: state of the science, current research needs, and future prospects". Journal of the Air & Waste Management Association. 64 (11): 1232–1250. doi:10.1080/10962247.2014.952846. PMID 25509545.
- ^ Anglada, Josef M.; Solé, Albert (November 2017). "The Atmospheric Oxidation of HONO by OH, Cl, and ClO Radicals". The Journal of Physical Chemistry A. 121 (51): 9698–9707. Bibcode:2017JPCA..121.9698A. doi:10.1021/acs.jpca.7b10715. PMID 29182863.
